Climate and Disease: Can the Environment Predict Transmission Risk?
- Emily Evenden, The George Washington University
- Nov 20, 2018
- 16 min read
Introduction: Climate and Disease
In my home state of Pennsylvania, we are again experiencing a hotter-than-average summer. Additionally, for the last month, this area has been inundated with storms which have dumped massive amounts of rain and caused significant flooding. Though a bad period of storms may be difficult to pin on climate change directly; consistent, increasing temperatures are surely a sign that our local environment is changing. This theory is corroborated by research conducted by the Pennsylvania State University which determined that Pennsylvania as seen a 1 degree Celsius increase in average temperature over the last century, as well as an increase in “wet” months. These trends are expected to continue more rapidly over this century [1]. In the future, continued environmental changes – more heat and more rain – will lead to new challenges in Pennsylvania, particularly in the field of public health. ![endif]--
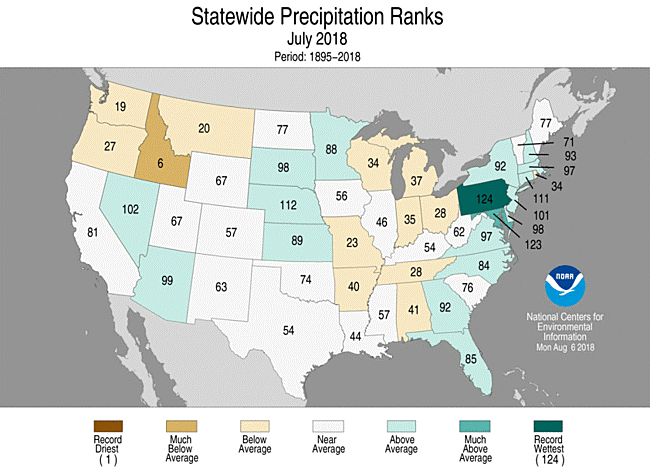
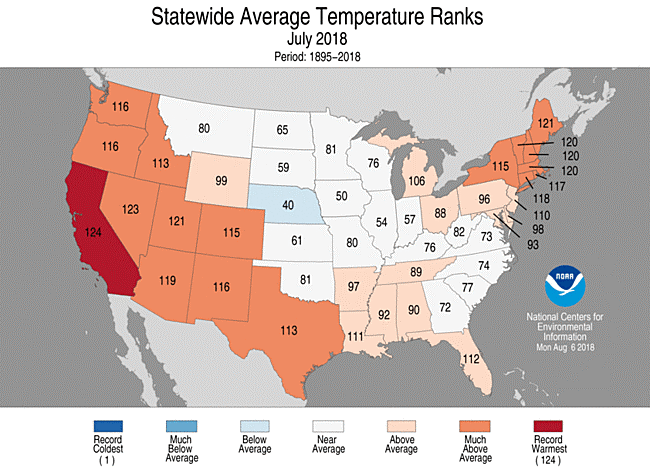
Pennsylvania experienced its wettest summer July on record with 7.37” of rain, up by 170% from its July average of 3.18”.The state has also been experiencing above-average temperatures, this month’s high at 96 degrees F. Larger, more frequent storms and consistently higher temperatures are potential impacts of climate change in this area [2].
An expected consequence of climate change is more frequent and wide-spread pandemics and epidemics [3]. More specifically, the spread of zoonotic diseases; illnesses carried and transmitted by vector species such as raccoon, skunks, bats, ticks, mosquitos, fleas, etc.; could spread to new areas [4]. Previous studies have shown that heavy rainfall in particular is associated with outbreaks of zoonotic diseases around the world since flooded areas provide breeding grounds for insects and disrupt the predator-prey relationships which regulate insect numbers [3]. Generally speaking, as the climate warms, the habitats of vector species expand, bringing them into contact with more people [5]. As of now, some of the zoonotic diseases recorded in Pennsylvania are Lyme disease, Anaplasmosis, Rocky Mountain Spotted-Fever, West Nile Virus, Rabies, Influenza, and Zika Virus [4]. Though the diseases listed above have already occurred in Pennsylvania, we could see the number of cases increase as our local environment gets warmer and wetter. Additionally, new diseases may arrive in our state as the climate becomes suitable for different invasive vector species.

This image depicts reported cases of four tick-borne diseases in the U.S. in 2011. Pennsylvania reported cases of all four disease, especially Lyme disease. Ticks are the most prominent vector in Pennsylvania [6].
Zoonotic Disease Spread
To predict how climate will impact the spread of disease, first we must understand how disease spreads normally. Zoonotic diseases are defined as “complex association between at least two organisms: pathogen and host” [7]. Traditional epidemiology simplifies the spread of disease to three pillars: a reservoir, a susceptible host, and the environment. A reservoir is the organism or ecosystem in which a pathogen normally breeds and multiplies. Often reservoirs do not show signs of illness despite housing the pathogen. Meanwhile, a susceptible host does not normally host the pathogen, meaning it will show signs of illness once infected and as the pathogen breeds. Finally, the environment describes the modes of transmission and the factors which bring reservoirs and hosts in contact with one another. In our case, transmission occurs through vector species, animals separate from reservoirs who carry the disease to uninhabited places. Together, these pillars are known as the chain of infection [8]. An example of the chain of infection is seen with Lyme disease. The reservoir of Lyme’s disease is a bacteria known as Borrelia burgdorferi. In the Northeast, this bacteria is often carried by a vector species, deer ticks. If a deer tick bites a human, the susceptible host, it will transmit the bacteria into our blood-stream and cause a bulls-eye rash [9]. When considering how zoonotic disease will be impacted by climate change, it is worth examining how climate effects all pillars in the chain of infection.

The diagram above explains the “chain of infection.” Typically, pathogens move from reservoirs to susceptible hosts via modes of transmission, such as vector species [8].
The Role of Ecological Niche
The geographic distribution of zoonotic disease outbreaks is linked to the geographic distribution of its chain of infection. In the field of disease biogeography, scientists study the spread of disease via the ecological requirements of pathogens, reservoirs, vectors, and hosts. In simple terms, all species occupy a niche, or a set of environmental requirements (climatic, biological, and behavioral) which allow a species to survive long-term [7]. Often the different species associated with a zoonotic disease have overlapping niches, allowing them to interact with each other. In disease biogeography, niche can be used to identify the potential geographic range of a disease based on the climate and biological conditions its chain of infection occurs in.
Niche can be further divided into a fundamental niche and a realized niche. The fundamental niche of a species is the total area with suitable environmental variables for long-term survival. The realized niche is the more limited area occupied by the species [7]. If a species expands from its realized niche to occupy more of its fundamental niche, it will appear in new places and grow in population size. In theory, zoonotic disease outbreaks are limited by the niche conditions constraining its chain of infection. In the context of climate change, a changing environment may expand the niches, both fundamental and realized, of zoonotic diseases and their associated species, allowing outbreaks in new areas. For example, in Pennsylvania, warmer and wetter weather may provide suitable living conditions for pathogen, reservoir, and vector species originally found further south. As the niche of these species moves further north, they will also expands north and could bring new disease with them. In particular, zoonotic threats to Pennsylvania under climate change include mosquito-borne West Nile Virus and Dengue Fever, which are not typical for the state [10].

This diagram demonstrates the difference between fundamental and realized niche. The fundamental niche encompasses all the environmental conditions in which a species can survive, while the realized niche includes the range of conditions a species currently occupies [11].
Using Niche to Map Disease Risk
Niche is a valuable tool for public health research because it can identify physical locations where a disease could exist, which allows us to focus disease prevention and treatment efforts. Previously, we have established that zoonotic disease-spread is linked the ecological niche of reservoir, vector, and host organisms; its chain of infection. With this knowledge, we can assume that areas specifically inhabited by these organisms may lead to greater risk of disease transmission. Therefore, if we can determine the general niche of the chain of infection, we can locate areas with higher transmission risk. This may sound simple, but determining the distribution of reservoir and vector species on a large-scale can be a tedious task if done on the ground. New geographic information system techniques have the potential to make this task faster and easier in a method called Ecological Niche Modeling.
[endif]--Ecological Niche Modeling links environmental characteristics and disease occurrence through computer modeling and statistical analysis. The purpose of this computerized method is to determine the range of abiotic environmental characteristics associated with the location of zoonotic disease cases. This method does not necessarily examine a specific species within a chain of infection, but instead identifies a general niche for outbreaks which can be used to identify other vulnerable areas based on similar environmental conditions. Modeling niche requires multiple data sources, including remotely-sense data and public health records, to make an all-encompassing model. During this process, disease occurrence data is overlaid on environmental data and analyzed statistically to determine what environmental conditions allow for disease transmission. By identifying a range of environmental conditions associated with disease, the model can then predict other areas capable of hosting the sickness [7]. ![endif]--

The image above shows the workflow of ecological niche modeling. Separate environmental and disease data are overlaid and combined. Together these datasets are analyzed mathematically in a modeling program to determine what environmental conditions are associated with disease presence. Then, using this established niche, the model identifies other areas suitable for hosting the disease [7].
Improving Disease Mapping
Mapping disease cases is a well-established technique for displaying and analyzing disease data. Easy to interpret, maps show how a disease is distributed over space. This method was first used by John Snow in 1854 when mapping instances of cholera in London [12]. Older methods of disease mapping and analysis focus on case density and proximity to determine the likelihood of transmission. Though these maps are useful, they are more practical for disease-surveillance than prediction because these statistics (disease density and proximity) do not consider the biological realism (necessary biological and environmental interactions) of disease spread. Simply put, zoonotic disease spread is not necessarily dependent on other human cases, nor does it spread linearly. Ecological Niche Modeling has the potential to map disease risk more realistically by considering the environmental variables suitable for pathogen survival. It also hopes to highlight areas with similar areas which may host a zoonotic disease but where health records are scarce. By analyzing disease occurrence in light of environmental variables, this method provides a new way to understand transmission [7].

This is the first known disease map, created by John Snow in 1854. Each black bar indicates a case of cholera at one address. The dot labeled “pump” indicates what Snow determined the source of the outbreak [12].
Methodology
Environmental Data
The environmental data used for Ecological Niche Modeling is typically remotely-sensed through satellites such as MODIS, a NASA satellite continuously collecting information about the Earth’s land surfaces, oceans, and atmosphere [13, 14]. Remotely-sensed data is distributed through both private and open-source means such as the commonly cited WorldClim free database [7, 15]. The environmental variables chosen for a study depend on the geographic scale of the project. For example, country or continental-scale studies might use variables such as temperature, precipitation, elevation, etc. which are measured continuously over large areas. However, if the research extent is smaller, perhaps over a province or county, a researcher may look into other variables such as standing water, or habitat fragmentation by roads, information which is not readily available on a large scale and nor very influential for general global trends [7].
Disease Data
In addition to climate, Ecological Niche Modeling requires disease occurrence data in order to properly correlate which environmental variables are associated with transmission. Disease records are obtained from many sources including government health institutions, online databases, or sampling in the field [13, 16, 17]. It is very important that cases are georeferenced properly, meaning their location is accurately recorded, since it will greatly impact the models’ accuracy. One benefit of Ecological Niche Modeling is that many modeling programs only require presence data (records of disease-positive locations) rather presence-absence data (records of disease-positive and disease-negative locations) which is much harder to obtain with certainty [7].
Statistical Modeling & Computational Analysis
Together, disease and environmental data are input to a computer model in order to analyze which environmental variables may predict disease transmission. Modeling can be done via many free, open-source programs including the Maximum Entropy Algorithm (MaxEnt), Niche Analyst (NicheA), BioClimatic Envelop Algorithm (BIOCLIM), Ecological Niche Factor Analysis (ENFA), Generalized Linear Models (GLM), Generalized Additive Models (GAM), etc. However, these models differ on whether they require presence-only or presence-absence data [7, 18].
Modeling programs utilize regression analysis to derive which environmental variables correlate with disease occurrence. This is done by first overlaying environmental and disease data, then rasterizing them. Rasterizing simply means dividing the data into a grid where each cell averages the data values within it, creating a simplified data set. Since disease is often reported haphazardly, occurrence is simplified to “yes or unknown” within its raster cell, identifying whether disease occurred within this area or not. Each raster cell contains averages of the chosen climate variables and whether disease has certainly occurred within that area.

The locations of a cutaneous leishmaniosis world-wide[7]. Mapping disease occurrence and removing outliers are the first steps of ecological niche modeling.
With this information, models can be used to conduct a regression analysis, a statistical technique used to determine independent and dependent variables [19]. In this case, the climate variables (e.g. temperature, precipitation, altitude, etc.) are potential independent variables, and disease occurrence is the dependent variable. The accuracy of regression analysis can be controlled and evaluated in several ways, including variable penalization and statistical measures of accuracy such as Receiver Operating Characteristic curve (AUC) and Kappa [7]. Proving accuracy is a hotly contested subject within Ecological Niche Modeling research, specifically in the topic of model validation. Often times the same data used to calculate which environmental variables correspond to disease is used to validate the same information, ensconcing bias into the model. This can be remedied by running the model on different locations or years than the original data used to build the models [7, 13].

Statistical analysis is used to identify what environmental conditions are associated with disease, in this case cutaneous leishmaniosis from the image above. In this photo, all values of temperature and precipitation for each raster cell were graphed. Those highlighted in red also contained disease. Based on this image, we can tell disease is limited to a specific range of temperature and precipitation [7].
Once the suitable range of variables is determined, the model highlights all cells with similar conditions, identifying areas where disease transmission is possible. The cells identified can be converted back into a map, displaying vulnerable areas in a more visually appealing manner. The end product displays the niche of the disease.

Once regression analysis determines the range of environmental variables correlated with disease, the now determined niche can be applied on a larger-scale to identify new areas at risk of transmission. This image displays the environmental suitability for cutaneous leishmaniosis globally. Red indicates extremely environmentally suitable areas [7].
Incorporating Climate Change
The methods described above explain how the initial niche of disease is determined under the present climate conditions. This process is essential for determining the future range of disease because it identifies the disease’s ideal environmental conditions. In order to apply this information to the future, we must establish how the environmental conditions may change. This has already been done by the International Panel on Climate Change, the global authority on climate forecasting and policy. A number of potential climate scenerios have been established by the IPCC based on emissions trends, economic development, land-use changes, etc [20]. The impact of these climate scenerios has also been quantified and converted into datasets, similar to remotely-sensed environmental data, which can be input to modeling software. In the case of Ecological Niche Modeling, once a niche is established, it can be applied to a model of potential climate conditions (temperature, rainfall, etc.) and identify the potential future geographic expanse of a disease, such as new areas which may be at risk due to climate change [21, 22].

Depicted above are the carbon dioxide emission scenarios established by the International Panel on Climate Change [20].
Case Study: Is Zika a Future Threat to Pennsylvania?
In 2015, the World Health Organization declared an outbreak of Zika virus a global health emergency as it spread from South America through Central America into the United States. The Zika virus is difficult to identify since it presents as cold symptoms and up to 80% of infected persons are asymptomatic. Though Zika is not necessarily a threat to healthy adults, this virus has extreme consequences when passed from pregnant mothers to their fetuses causing birth defects, primarily microcephaly [23, 24]. During the 2015 pandemic, cases of microcephaly in newborns grew twenty-fold in Brazil [24]. Because little was known about Zika virus, towns as far north as New York City began to prepare for its potential spread, creating an urgent need to learn more about the virus. Due of this uncertainty, scientists are trying to quantify the global risk of Zika using ecological niche modeling and assess future risk using climate models.
In a study sponsored by the University of California Berkeley and the University of KwaZuluNatal, researchers attempted to quantify the niche of Zika using Ecological Niche Modeling. Once Zika’s potential niche was determined, it was compared with the modeled niche of the more common and closely-related zoonotic disease, Dengue Fever. The aim of this comparison was to determine whether the niche of Dengue Fever could be used to estimate the potential range of Zika. Additionally, the researchers compared the modeled niches of Dengue Fever and Zika with similarly modelled niches of the diseases’ three mosquito vector species (Aedes aegypti, Ae.africanus, and Ae.albopictus) to determine whether vector range correlates with Dengue Fever or Zika outbreaks. In other words, the second comparison between disease and vector niches was meant to see if vector range is the primary limiting factor for Dengue Fever and Zika spread. This is very valuable information for areas, like Pennsylvania, that are home to some of the vector species, but do not have outbreaks of either disease. Finally, all five modelled niches (Dengue Fever, Zika virus, and the three mosquito species) were then applied to a climate model (accessed through WorldClim) to predict the potential range of Zika under changing climate conditions [24]. Because Zika could spread further into North America with climate change, it is important to know what areas are at risk in order to prepare for future outbreaks.

Image B displays the potential geographic distribution of Dengue Fever based on its niche, while Image C displays the same for Zika virus. Based on this study, Dengue Fever has a significantly broader niche than Zika virus [24].
First, scientists modeled the ecological niche of Zika and Dengue Fever to confirm whether past dengue outbreaks may indicate the potential expanse of the Zika virus. This was done using occurrence data of both diseases ranging from 1947 to 2000 (omitting the 2015 Zika outbreak) as well as data from 19 WorldClim variables (not detailed in the article). Together, this information was run through ten different statistical modeling programs (Maxent, GLM, GAM, etc.) to find the most accurate model. The same method was used to determine the niche of the three mosquito species. Once conducted, the results showed the Dengue Fever and Zika, though related, have very different niches (Zika remaining more strictly tropical). As a result, historical Dengue Fever outbreaks cannot be used to determine the potential range of Zika. Additionally, neither Dengue Fever nor Zika outbreaks strictly follow the niches of its mosquito vectors, meaning it is not guaranteed to occur in places where its vector exists. This means that the actual range, or realized niche, of both diseases may be limited by something other than its vectors’ ranges [24].
Additionally, the niche profiles of Zika and its mosquito vectors were applied to the chosen climate model to predict the future geographic area at risk of Zika transmission. This study found that Zika will only be consistently present in the southeast region of the U.S. under this specific climate scenario. Additionally, based on the range of Zika’s vector mosquitos, there is potential for it to spread further up the East Coast of the U.S. and small areas of the West Coast [22]. It’s important to note that that this ecological niche model is more thorough, but conservative, compared to other niche models of Zika. Something this study lacks is that it does not include explanatory variables such as socioeconomic status, which may play a large role in how a disease advances due to inequalities in health resources and access. Therefore, the distribution of Zika itself could be larger in the future than what is shown here and its advance may be uneven.

Images A and B display the current potential range of Dengue Fever (blue) and Zika virus (red), along with the current potential range of their three mosquito vectors. Images C and D show the same ranges quantified using a future climate scenario from the WorldClim database. According to the model, climate change will expand the niche of all three mosquito vectors, as well as Dengue Fever and Zika virus [24].
Based on this study, is Zika virus a threat to Pennsylvania? According to this particular study, it is unlikely. Though Pennsylvania is home to some of the vector species known to carry Zika virus in other areas, it is unlikely that state’s climate, even considering climate change, will support Zika virus in the long-term. However, what we can learn from this study is that model accuracy and decisions during the scientific process can seriously impact outcomes and real-time decision-making. In this case, the decision to exclude data from the 2015 Zika outbreak may have made the niche model either more conservative than actuality or more realistic than previous models, it’s difficult to know for sure. The results of this model, if used for decision-making, may save local, state, and federal governments in North America billions of dollars in unnecessary environmental and medical surveillance and treatment efforts, or lead to gaps in disease prevention. Though this particular example of ecological niche modeling may not lead to dramatic results in Pennsylvania, it demonstrates how this method may be used in the future for other diseases.
Since ecological niche modeling is a relatively new methodology, especially in public health research, the papers published sparsely cover a wide variety of topics including human diseases, crop blights, and invasive species. A concurrent emerging branch of research includes modeling the spread-rate and dispersal methods of diseases and invasive species both now and under future climate conditions. There is also lot of potential for incorporating animal behavior, and other small-scale interactions, into zoonotic disease models. For Pennsylvania specifically, other zoonotic diseases of interest include West Nile Virus, Dengue Fever, and potentially Malaria, all of which have very limited publications about their potential impacts on the Northeast region. Ecological Niche Modeling is a promising technique for public health research, but requires further refining and application in order to determine its full predictive potential. If anything, it is an interesting diversion from older methods, incorporating the biological realism of disease spread.
Emily Eveden is a student at the George Washington University is Washington, D.C. and a former USAID Geocenter Virtual intern. She has previously contributed to the YouthMappers blog, find her work here.
Citation
[1] Shortle, James, et al. Pennsylvania Climate Impact Assessment Update. The Pennsylvania State University, 2015, p. 6, Pennsylvania Climate Impact Assessment Update.
[2] NOAA National Centers for Environmental Information, State of the Climate: National Climate Report for July 2018, published online August 2018, retrieved on August 14, 2018 from https://www.ncdc.noaa.gov/sotc/national/201807.
[3] Cho, Renee. “How Climate Change Is Exacerbating the Spread of Disease.” State of the Planet, Columbia University Earth Institute , 10 Dec. 2014, www.blogs.ei.columbia.edu/2014/09/04/how-climate-change-is-exacerbating-the-spread-of-disease/
[4] Deese, Brian, and Ronald A. Klain. “Another Deadly Consequence of Climate Change: The Spread of Dangerous Diseases.” The Washington Post, WP Company, 30 May 2017, www.washingtonpost.com/opinions/another-deadly-consequence-of-climate-change-the-spread-of-dangerous-diseases/2017/05/30/fd3b8504-34b1-11e7-b4ee-434b6d506b37_story.html?utm_term=.de570a741e48.
[5] National Center for Emerging and Zoonotic Infectious Diseases (NCEZID). “Information about Pennsylvania in NCEZID Publications (2011–Present).” Centers for Disease Control and Prevention, Centers for Disease Control and Prevention, 31 Aug. 2017, www.cdc.gov/ncezid/stories-features/browse/states/pennsylvania.html.
[6] Centers for Disease Control and Prevention (CDC). National Center for Emerging and Zoonotic Infectious Diseases: our work, our stories, 2011-2012. Atlanta, GA: CDC; 2013. https://www.cdc.gov/ncezid/pdf/annual-report.pdf#page=79.
[7] “Principles of Epidemiology in Public Health Practice - Lesson 1.” Centers for Disease Control and Prevention, Centers for Disease Control and Prevention, 18 May 2012, www.cdc.gov/ophss/csels/dsepd/ss1978/lesson1/section10.html.
[8] National Center for Emerging and Zoonotic Infectious Diseases (NCEZID) Division of Vector-Borne Diseases (DVBD). “Lyme Disease Transmission.” Centers for Disease Control and Prevention, Centers for Disease Control and Prevention, 4 Mar. 2015, www.cdc.gov/lyme/transmission/index.html.
[9] Escobar, Luis E., and Meggan E. Craft. “Advances and Limitations of Disease Biogeography Using Ecological Niche Modeling.” Frontiers in Microbiology, vol. 07, 2016. Directory of Open Access Journals, doi:10.3389/fmicb.2016.01174.
[10] Haq, Aliya. NRDC Fact Sheet: Climate and Health in Pennsylvania. NRDC Fact Sheet: Climate and Health in Pennsylvania, National Resource Defense Council, 2015.
[11] Mikell, Meredith. “Competitive Exclusion Principle: Definition & Example.” Study.com, Study.com, www.study.com/academy/lesson/competitive-exclusion-principle-definition-example.html.
[12] Mapping the 1854 London Cholera Outbreak. University of Delaware, www1.udel.edu/johnmack/frec682/cholera/.
[13] Escobar, Luis E, et al. “Ecological Approaches in Veterinary Epidemiology: Mapping the Risk of Bat-Borne Rabies Using Vegetation Indices and Night-Time Light Satellite Imagery.” Veterinary Research, vol. 46, no. 1, 2015, doi:10.1186/s13567-015-0235-7.
[14] Maccherone, Brandon. “About MODIS.” MODIS Moderate Resolution Imaging Spectroradiometer, National Aeronautics and Space Administration, www.modis.gsfc.nasa.gov/about/.
[15] Elith, Jane, et al. “Novel Methods Improve Prediction of Species' Distributions from Occurrence Data.” Ecography, vol. 29, no. 2, 1 Apr. 2006, pp. 129–151.
[16] Meentemeyer, Ross K., et al. “Early Detection Of Emerging Forest Disease Using Dispersal Estimation And Ecological Niche Modeling.” Ecological Applications, vol. 18, no. 2, Mar. 2008, pp. 377–390., doi:10.1890/07-1150.1.
[17] Colacicco-Mayhugh, Michelle. “Applications of Ecological Niche Modeling to Enhance Medical Threat Assessment and Disease Control and Prevention Strategies.” U.S. Army Medical Department Journal, 2009, pp. 28–32.
[18] Bradie, Johanna, and Brian Leung. “A Quantitative Synthesis of the Importance of Variables Used in MaxEnt Species Distribution Models.” Journal of Biogeography, vol. 44, no. 6, June 2017, pp. 1344–1361.
[19] “Regression Analysis.” Wikipedia, Wikimedia Foundation, 13 July 2018, www.en.wikipedia.org/wiki/Regression_analysis.
[20] IPCC Special Report: Climate Scenarios. International Panel on Climate Change, 2000, IPCC Special Report: Climate Scenarios, www.ipcc.ch/pdf/special-reports/spm/sres-en.pdf.
[21] Beck, Jan. “Predicting climate change effects on agriculture from ecological niche modeling: who profits, who loses?” Climate Change, vol. 116, 2013, pp. 177-189., doi: 10.1007/s10584-012-0481-x.
[22] Gonzalez, Camila, et al. “Climate Change and Risk of Leishmaniosis in North America: Predictions from Ecological Niche Models of Vector and Reservoir Species.” Neglected Tropical Diseases, vol. 4, no. 1, Jan. 2010. doi:10.1371/journal.pntd.0000585.
[23] “Zika Virus.” Centers for Disease Control and Prevention, Centers for Disease Control and Prevention, 1 May 2017, www.cdc.gov/zika/symptoms/symptoms.html.
[24] Carlson, Colin J., et al. “An Ecological Assessment of the Pandemic Threat of Zika Virus.” PLOS Neglected Tropical Diseases, vol. 10, no. 8, 2016, doi:10.1371/journal.pntd.0004968.
![endif]--




Comments